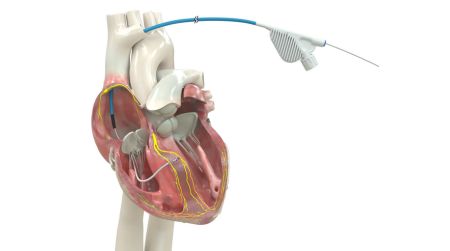
March 23, 2026 – Medtronic plc has acquired approval from the U.S. Meals and Drug Administration (FDA) for an expanded indication for the OmniaSecure™ defibrillation cable. The lead is now permitted for placement within the left bundle department space (LBB) and can be utilized for conduction system stimulation (CSP), which carefully mimics the pure physiology of the guts. Moreover, sufferers requiring cardiac resynchronization could profit from left bundle branch-optimized cardiac resynchronization remedy (LOT-CRT), a novel remedy that mixes CSP with left ventricular pacing to additional enhance affected person outcomes.
The OmniaSecure defibrillation lead connects to an implantable cardioverter defibrillator (ICD) or cardiac resynchronization remedy defibrillator (CRT-D) to deal with probably life-threatening ventricular tachyarrhythmias, ventricular fibrillation (VT/VF), and bradyarrhythmias. Along with being the primary defibrillation lead permitted for placement within the LBB area, the OmniaSecure lead is the smallest diameter defibrillation lead available on the market (4.7 French or 1.66 mm) and the one one permitted for adults and adolescent pediatric sufferers 12 years of age and older.
Proceed studying