Philips introduces the intervention platform IntraSight Plus
Philips has launched the IntraSight Plus coronary intervention platform, which has been permitted for scientific use in Europe and the US.
With U.S. Meals and Drug Administration (FDA) 510(okay) clearance and CE marking, the interventional steerage system goals to streamline entry to diagnostic and therapy planning instruments for physicians.

Uncover B2B advertising and marketing that delivers
Mix enterprise intelligence and editorial excellence to achieve engaged professionals throughout 36 main media platforms.
Extra data
Philips acknowledged that interventional cardiologists have historically solely relied on angiography for percutaneous coronary interventions (PCI), a minimally invasive process.
Present guideline-supported instruments, reminiscent of intravascular ultrasound and physiology, are sometimes delivered by way of separate techniques, which may hinder adoption in busy catheterization laboratories.
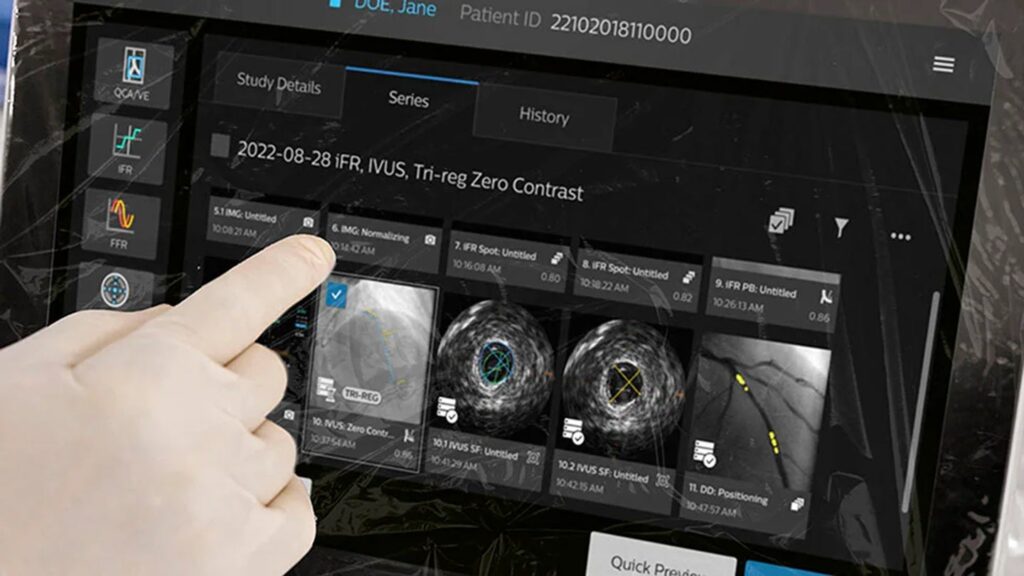
Philips’ IntraSight Plus goals to deal with these challenges by offering a single built-in resolution that may cut back system uptime by as much as 47% and help bedside operation.
Throughout this month, Dr. Jan-Malte Sinning the primary affected person with IntraSight Plus at Cellitinnen-Krankenhaus St. Vinzenz in Cologne, Germany. The hospital just lately upgraded its catheterization laboratory with Philips Azurion interventional suites.
The mixing of IntraSight Plus into this surroundings goals to enhance the workflow for medical doctors.
Utilizing each Azurion and IntraSight Plus, Dr. Sinning full revascularization earlier than discharge.
IntraSight Plus combines Philips’ present applied sciences, reminiscent of IntraSight and SyncVision, and gives options reminiscent of co-registration, instantaneous wave-free ratio physiology/fractional energy reserve (iFR/FFR), Class IA IVUS and real-time machine visualization inside one system.
Philips image-guided remedy gear enterprise chief Stacy Beske stated: “As extra superior expertise is built-in into the catheter laboratory, the necessity for connectivity between imaging instruments is extra essential than ever earlier than.
“IntraSight Plus was constructed on suggestions from our prospects and represents a elementary shift in interventional cardiology – a future pushed by built-in intelligence that permits physicians to deal with the decision-making that solely they will do.”
Beforehand, the FDA permitted Philips’ SmartHeart AI, offering physicians with a device that streamlines cardiac magnetic resonance workflows.
