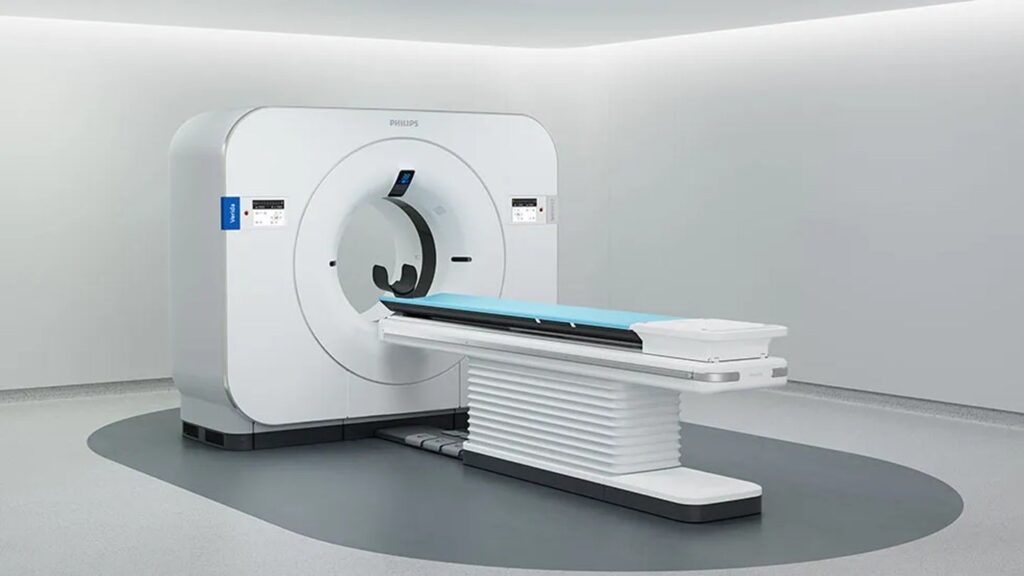
Philips receives FDA clearance for Verida spectral CT system
Philips has obtained 510(okay) clearance from the U.S. Meals and Drug Administration (FDA) for its Verida system.
The system is an AI-powered detector-based spectral computed tomography (CT) system developed to enhance diagnostic accuracy in medical functions.

Uncover B2B advertising and marketing that delivers
Mix enterprise intelligence and editorial excellence to succeed in engaged professionals throughout 36 main media platforms.
Extra data
Verida integrates Spectral Exact Picture know-how utilizing a third-generation Nano-panel Exact dual-layer detector that gives intrinsic noise discount and an AI-based deep studying reconstruction engine.
These options are meant to cut back noise and enhance picture high quality throughout CT imaging. Clinicians have the pliability to regulate picture noise to their preferences.
Verida permits uninterrupted spectral imaging primarily based on Philips’ detector-based spectral CT know-how, eliminating the necessity for separate scans or workflow modifications.
The system options enhanced options to help medical workflows, together with enhancements to the spectral outcomes era pipeline and up to date computing infrastructure for improved efficiency and usefulness.
It reconstructs 145 photos per second, making full exams obtainable inside 30 seconds and supporting as much as 270 exams day by day.
By capturing each high-energy and low-energy information in a single scan, clinicians obtain each standard and spectral outcomes for superior tissue characterization and materials differentiation, lowering the necessity for pre-selection or repeat scans.
Verida is appropriate for diagnostic imaging in interventional radiology, radiology, cardiology and oncology.
It’s indicated for functions to the top, complete physique, coronary heart and blood vessels in sufferers of all ages, and may also be used for low-dose CT screening for lung most cancers, in addition to for therapy preparation and radiation remedy planning.
Philips CT enterprise chief Dan Xu stated, “With the FDA approval for Verida, we’re bringing the subsequent evolution of spectral CT to extra markets. By combining always-on spectral imaging with AI-powered reconstruction, Verida permits physicians to see extra directly, supporting sooner, extra knowledgeable selections and increasing the function of CT throughout medical pathways.”
Final month, Philips obtained FDA approval for DeviceGuide, an AI-based software program designed to offer docs with real-time visible help throughout mitral valve restore procedures.
