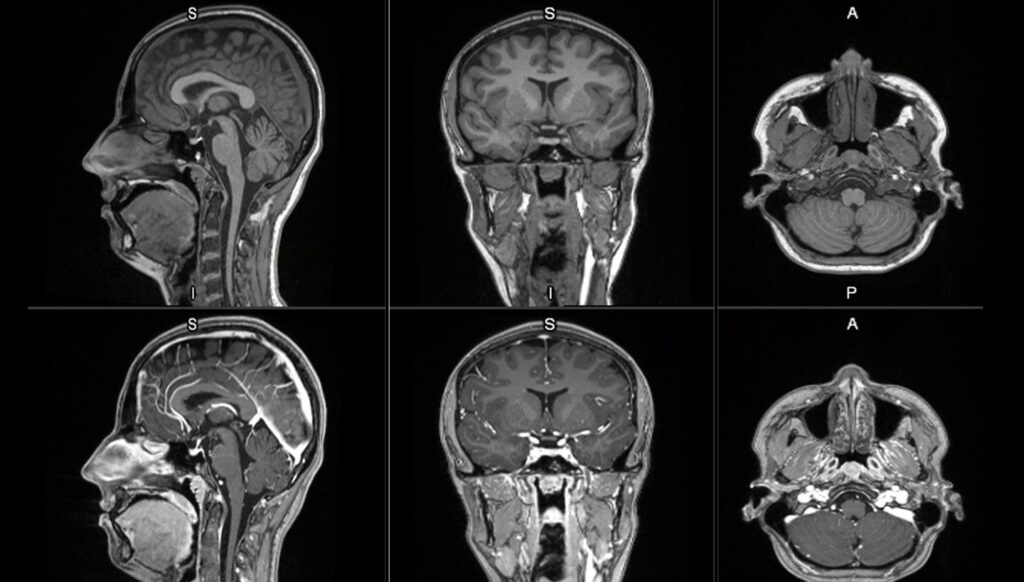
First affected person to obtain a dose in GE HealthCare’s Part II/III LUMINA scientific trial
GE HealthCare has dosed the primary affected person in its worldwide, multicenter Part II/III LUMINA scientific trial for the manganese-based magnetic resonance imaging (MRI) distinction agent, mangaciclanol.
The trial started on the Mayo Clinic in Rochester, Minnesota, US, and focused adults and pediatric sufferers aged two years and older, following accelerated designation by the US Meals and Drug Administration (FDA).

Uncover B2B advertising that delivers
Mix enterprise intelligence and editorial excellence to achieve engaged professionals throughout 36 main media platforms.
Extra data
If authorised, mangaciclanol might function an alternative choice to gadolinium-based MRI distinction brokers presently in use worldwide. The FDA’s fast-track designation goals to speed up analysis of therapies that deal with severe unmet wants.
The brand new distinction agent is meant to enhance MRI detection and visualization of lesions with irregular vascularity within the central nervous system and physique.
MRI distinction brokers assist enhance the visibility of irregular buildings throughout scans, supporting higher differentiation between wholesome and unhealthy tissue.
Early scientific photographs of mangaciclanol point out comparable relaxivity to gadobutrol, a generally used gadolinium-based agent, with related diagnostic potential.
In contrast to gadolinium, a uncommon earth metallic, manganese is a vital ingredient in meals that’s endogenously regulated within the human physique, and the macrocyclic construction of mangaciclanol is designed to restrict retention.
Distinction brokers are needed in nearly a 3rd of MRI procedures worldwide, with an estimated 65 million gadolinium-enhanced examinations carried out yearly.
The provision of gadolinium is extremely depending on mining actions in China, which poses a possible provide threat. Nonetheless, manganese is extra extensively obtainable from sources in Australia, Gabon and South Africa, mitigating such dangers.
The agent might additionally deal with environmental considerations as manganese is of course current in water sources, probably lowering the influence of excreted distinction brokers on groundwater.
Peter Arduini, president and CEO of GE HealthCare, mentioned: “As demand for diagnostic imaging continues to rise, we proceed to broaden our imaging pipeline to higher meet affected person wants.
“This scientific milestone builds on GE HealthCare’s management in distinction media, as mangaciclanol has the potential to rework the MR imaging market and strengthen provide chain resilience.”
The Part I trial outcomes of Mangaciclanol confirmed that the drug was effectively tolerated, with no severe opposed occasions, dose-limiting toxicities, or clinically vital findings noticed.
GE HealthCare’s Pharmaceutical Diagnostics division is a acknowledged supplier of imaging sources, supporting 140 million affected person procedures yearly.
GE HealthCare distinction media have been utilized in MRI, ultrasound and X-ray/CT imaging for greater than 40 years.
Mangaciclanol stays in scientific growth and has not but been authorised for industrial use.
Final week, GE HealthCare expanded its partnership with RadNet subsidiary DeepHealth to broaden entry to AI-powered breast most cancers screening options.
