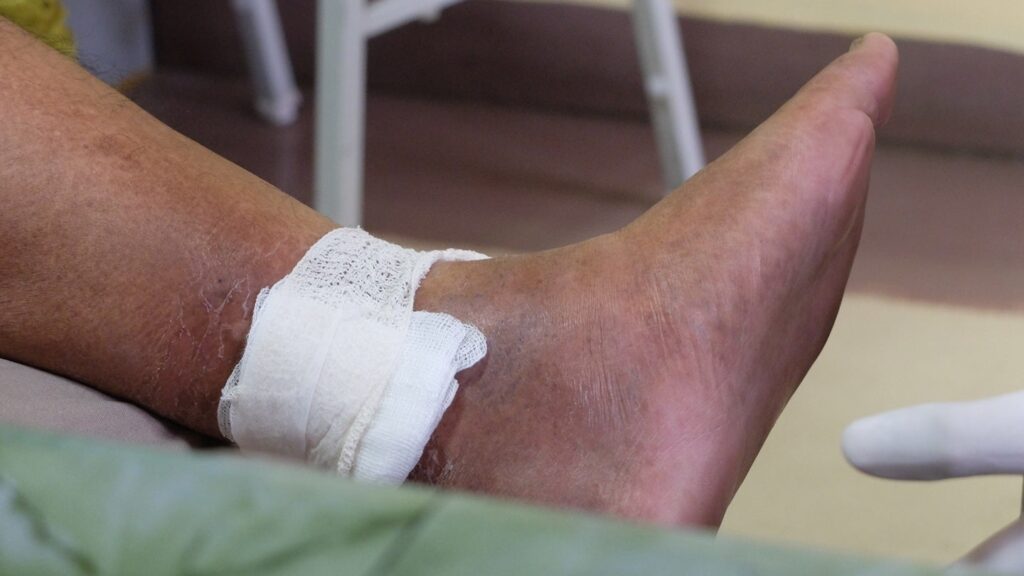
enVVeno Medical Obtains FDA IDE Clearance for Venous Valve Analysis
enVVeno Medical has obtained approval from the US Meals and Drug Administration (FDA) for a pivotal examine of a non-surgical substitute venous valve.
The approval permits enVVeno Medical to start pivotal analysis into the enVVe System’s transcatheter venous valve endoprosthesis (TAVVE), aimed toward treating extreme deep persistent venous insufficiency (CVI).

Uncover B2B advertising and marketing that delivers
Mix enterprise intelligence and editorial excellence to succeed in engaged professionals throughout 36 main media platforms.
Extra data
Deep venous CVI, normally on account of deep vein thrombosis, results in blood pooling and elevated venous stress within the legs.
This milestone allows the development of a possible remedy for almost three million U.S. sufferers with extreme CVI brought on by dysfunctional valves in deep leg veins.
These sufferers presently have restricted efficient remedy choices, and the examine will examine a minimally invasive strategy.
Robert Berman, CEO of enVVeno Medical, stated: “Now we have labored very carefully with the FDA to make sure that the TAVVE examine is structured to ship every part the FDA desires to see when evaluating the security and efficacy of enVVe, and that we’re absolutely aligned.
“A transcatheter-based non-surgical substitute venous valve has at all times been thought-about the ‘holy grail’ for sufferers with extreme CVI, and curiosity and enthusiasm amongst physicians and their scientific groups has been extraordinarily excessive.
“We’re excited to formally start choosing and activating scientific websites and look ahead to the primary implant process utilizing the enVVe system.”
The TAVVE examine will start later this 12 months with an preliminary cohort of ten sufferers, whose 30-day security outcomes will probably be despatched to the FDA for overview. This group will proceed to be monitored and their progress will probably be shared publicly.
The second part will enroll 220 sufferers: 165 will obtain the enVVe valve and 55 will probably be randomized to straightforward care throughout as much as 40 scientific websites within the US.
The examine entails interventional radiologists, vascular surgeons and interventional cardiologists.
Key options of the enVVe system embrace a minimally invasive process, a self-expanding biocompatible nickel-titanium alloy framework, three valve sizes, a mono-cusp leaflet of porcine pericardium, and a compact 13-Fr supply profile.
In April 2024, enVVeno Medical introduced optimistic topline efficacy knowledge from its SAVVE scientific trial investigating the VenoValve, displaying that 94% of sufferers utilizing the valve confirmed scientific enchancment.
