
Abbott receives FDA approval for Ultreon 3.0 imaging platform
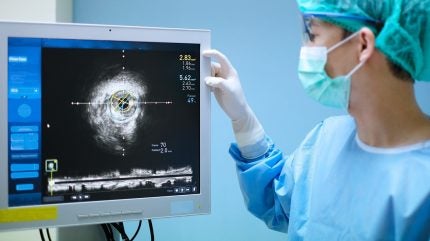
Abbott has acquired U.S. Meals and Drug Administration (FDA) approval and CE Mark approval for its Ultreon 3.0 AI-powered coronary imaging platform.
The approval permits the combination of automated imaging insights with real-time planning steering throughout percutaneous coronary intervention (PCI) procedures to help physicians and probably enhance affected person outcomes.

Uncover B2B advertising that delivers
Mix enterprise intelligence and editorial excellence to achieve engaged professionals throughout 36 main media platforms.
Extra data
The subsequent technology Ultreon 3.0 makes use of AI with optical coherence tomography (OCT) to ship high-resolution, real-time imaging throughout minimally invasive procedures that deal with clogged coronary heart arteries.
The system is designed to help a completely built-in scientific workflow to allow sooner, correct coronary interventions.
A characteristic of the Ultreon 3.0 software program is the one-second OCT pullback, a quick, infrared light-based imaging approach.
It captures detailed cross-sections of coronary arteries whereas providing larger decision than intravascular ultrasound (IVUS) and the potential to be used with little or no distinction.
Lowering the quantity of distinction could also be particularly useful for sufferers with kidney illness, which accounts for about 25% of these with coronary artery illness.
Key capabilities of Ultreon 3.0 embody detailed assessments of the scale, form and placement of the blockage to help correct stent placement, in addition to post-procedure assessments to verify improved blood movement.
The system encompasses a streamlined design, superior AI-driven workflow effectivity and the flexibility to effectively plan procedures for complicated coronary instances.
Ethan Korngold, Chief Medical Officer of Abbott Vascular Enterprise, stated: “Complemented by our industry-leading coronary portfolio, Ultreon 3.0 makes OCT imaging extra intuitive and highly effective for physicians, reimagining imaging as a software for exactly guided interventions earlier than and after procedures.
“Advances like Ultreon 3.0 reveal Abbott’s management in AI-powered imaging and as a trusted associate in delivering smarter, extra personalised coronary care.”
Ultreon 3.0 builds on the earlier model by introducing enhanced AI capabilities centered on the wants of complicated coronary PCIs.
Final month, Abbott introduced the combination of its Precision Oncology portfolio into Flatiron Well being’s cloud-based digital medical document (EMR) platform, OncoEMR.
